The Academy Is Your “Help Button” in Practice and Patient Care
The Academy is leading advocacy action on a number of legislative and regulatory concerns.
Most of our members are well aware of the Academy’s advocacy efforts on their behalf that involve both legislative and regulatory concerns and include some of the following:
- Monitoring and giving a voice to the specialty for the Centers for Medicare & Medicaid Services (CMS) policies that infiltrate daily practice and patient care
- Challenging private payer policies that affect payment and practice workflow
- Organizing and leading action against state legislative efforts centering around scope of practice
- Spearheading the creation of new CPT codes and their subsequent valuation through the AMA CPT/RUC process
 James C. Denneny III, MD
James C. Denneny III, MD
AAO-HNS/F Executive Vice President and CEO
The direct relationship between policies and rules that are perceived as burdensome, inappropriate, and unnecessary as well as the lack of necessary equipment and supplies and the overall wellness of the physician community has been established multiple times over the last five to 10 years. The Academy has identified a number of areas of high importance to our members and their patients that we have taken on and made a difference.
The Joint Commission
By mid-2018 the increasing burden of policies concerning rules for hospitals and clinics by The Joint Commission was negatively affecting patient care. Otolaryngologists were not immune from this phenomenon and were near the front of the line when it came to the “danger level” caused by this increasing series of both in-office and facility-based requirements related to instrument cleaning, packaging, and storage.
To identify and clarify the specific issues and concerns affecting our members and their patients, we sent out a concise survey of six questions through multiple distribution avenues. We received 158 detailed responses in the first 10 days.
This led to a meeting with The Joint Commission during which we discussed the results of the survey and major areas of concern of our members. The Joint Commission addressed each of these issues individually. The evidence we were able to quickly collect from members and judiciously present to The Joint Commission resulted in significant alterations of the major policies negatively affecting our members.
COVID-19
I received a phone call from a member in mid-March 2020 just as COVID-19 was taking off in the United States who asked me if any members were reporting anosmia in COVID patients. There were a few isolated reports circulating around worldwide, but no known link had been firmly established. I contacted the Patient Safety and Quality Improvement (PSQI) Committee, and we created the “Anosmia Reporting Tool” and publicly deployed it in late March. This resulted in early media coverage by NPR, The New York Times, USA Today, Bloomberg, and other national and international print, digital, and TV coverage. A paper was published in the AAO-HNSF journal Otolaryngology–Head and Neck Surgery with the results of the first 158 patients in April 2020. The tool was closed in 2021 with over 1,000 patients reported. A public statement, the reporting tool, and the journal article were instrumental in the Centers for Disease Control and Prevention (CDC) adding anosmia to the major symptom list of COVID-19 infection in April 2021.
As the U.S. transitioned back toward more widespread delivery of healthcare and otolaryngologists expanded their practice to include nonemergent and time-sensitive patient care, they were seeking consistent guidance and principles to reinstitute diagnostic and therapeutic interventions. The Academy, through the Future of Otolaryngology Task Force with the collaboration of the otolaryngology specialty societies, developed recommendations for a safe return to practice that proved to function as a living document giving guidance to physicians and their practices as the pandemic progressed. We were also active from the beginning of the pandemic communicating about and advocating for adequate personal protective equipment for the entire specialty.
Tracheostomy Tube Shortage
Last fall it came to our advocacy team’s attention that there was a severe shortage of tracheostomy tubes in the U.S., particularly at children’s hospitals. This triggered an immediate investigation into the situation in collaboration with pediatric otolaryngologists and the American Society of Anesthesiologists, which resulted in multiple conversations with the FDA and with both American and European suppliers. We found that this was a multifactorial supply-chain issue with no simple solution. We sent a formal letter to the FDA and collaborated with them in trying to identify additional supplies. Special thanks go to Julie L. Wei, MD, President of the American Society of Pediatric Otolaryngology, Soham Roy, MD, and Ken Kazahaya, MD, who provided us with instrumental data. Dr. Wei led the efforts to acquire critical data requested by the FDA through a survey completed by 130 pediatric otolaryngologists. Ultimately this led to the opportunity to create a joint statement on safe use and re-use of pediatric tracheostomy tubes.
This is an inspirational example of how a comprehensive, effective response to a critical problem can be addressed rapidly when affected stakeholders can work together to identify the full extent of a problem and help create a reasonable solution. It also highlights the benefit and need for all areas of otolaryngology to work effectively together to improve care.
Suction Tubing Shortage
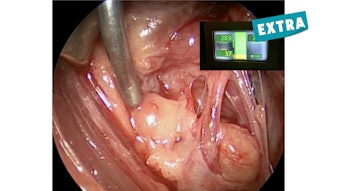
Earlier this year a group of our pediatric otolaryngologists informed me that they heard that the primary supplier was no longer able to provide flexible suction tubes for pediatric bronchoscopy and esophagoscopy rigid scopes. We initiated contact with our long-time colleague at that industry partner. The ensuing conversation resulted in understanding the regulatory change in Europe that led to the change and a detailed review of alternatives. For more on this, read the article by Dr. Wei and Dr. Roy in the June Bulletin EXTRA.
NIM Monitors
A chance meeting between an Academy past president at a recent meeting resulted in a conversation about problems they were having with the facial monitoring system’s software that was affecting the alarm system in the newest product. After alerting the FDA, we contacted the manufacturer and set up a series of meetings with the leadership of the American Neurotology Society, American Otological Society, our advocacy team, and their complete team. These discussions were taken very seriously, and an action plan was created within two weeks that included education materials for employees, hospital staff, and physician users and planned observation of selected surgeons to receive feedback on the product. This all started with someone who did not realize that the Academy got involved in this area of advocacy.
We have developed collegial, working relationships with CMS, the CDC, the FDA, our industry partners, and many of the private health insurers that allow us to work with our specialty societies to intervene on behalf of our members and patients in most areas affecting patient care. The earlier that we learn about your areas of concern, the more likely we can help!