Collaboration and Action to Address Supply Chain Shortages
Regardless of membership subspecialties, AAO-HNS leadership continues to leverage its strong voice and relationships to serve us and our collective patients.
Soham Roy, MD, MMM, and Julie L. Wei, MD, MMM

As the pediatric otolaryngology community voiced a need to address these shortages affecting children, we connected with James C. Denneny III, MD, Executive Vice President and CEO of the American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS). Dr. Denneny already had several meetings with the U.S. Food and Drug Administration (FDA) and the U.S. Department of Health and Human Services (HHS) representatives to highlight how the supply chain shortage was impacting hospitals, patients, and families and to explore possible options in collaboration with manufacturers. He not only welcomed the pediatric otolaryngology perspective and concerns, but he broadened the conversation and included both of us in those meetings.
A small American Society of Pediatric Otolaryngology (ASPO) task force was convened to provide data regarding the number of children impacted. The task force quickly designed and deployed a national survey asking one representative from each children’s hospital or practice to respond and then provided summarized data to the FDA on estimated number of children impacted. Ultimately this led to the opportunity to create a joint expert consensus statement on safe use and re-use of pediatric tracheostomy tubes.
The FDA shared this information with the public and facilitated critical communication with manufacturers. ASPO also shared this statement as well as the survey results with its membership, providing awareness, education, and allowing this statement to be widely disseminated to patient families and caretakers.
In recent weeks, otolaryngologists and particularly pediatric otolaryngologists encountered a new supply chain shortage issue—flexible suction catheters commonly used in rigid endoscopy for diagnostic or therapeutic airway procedures were discontinued from manufacturing. Dr. Roy and Dr. Denneny reached out to Karl Storz, the manufacturer of this flexible suction catheter, to ask for guidance and alternate product recommendations.
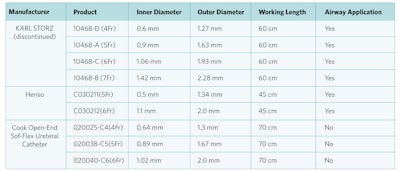
On behalf of the AAO-HNS and ASPO, we are grateful to Karl Storz leadership and representatives for their comprehensive search and summary provided on alternate products (Table 1). The company provided reliable information on the country and manufacturer name of alternate products, as well as available data on how products are used. Karl Storz also shared references for two studies1,2 that described the use of non-bronchial suction catheters for intended pediatric endobronchial/endotracheal use to further support appropriate use of alternate products given their suction catheter product will no longer be manufactured.
ASPO is grateful for the support from the AAO-HNS and the opportunities to collaborate and address myriad issues affecting our patients and members such as the supply chain, advocacy, and reimbursement. The benefits of AAO-HNS membership impacts our patients in ways that we haven’t imagined nor anticipated. Regardless of membership subspecialties, AAO-HNS leadership continues to leverage its strong voice and relationships to serve us and our collective patients.
Table 1. Alternate Product Information
References
- Kesan KV KS, Ade A, Kothari P. Use of ureteral catheter for under vision suctioning in rigid therapeutic bronchoscopy in children: A novel approach. Archives of Otolaryngology and Rhinology. 2020;6(3):71-75. doi:10.17352/2455-1759.000126
- Papoff P, Cicchetti R, Montecchia F, Midulla F, Ceccanti S, Cozzi D. Intentional tracheoesophageal fistula cannulation for gastric decompression in type C esophageal atresia. Paediatr Anaesth. Apr 2018;28(4):367-369. doi:10.1111/pan.13351