Reg-ent Moves to Phase II: Research—Clinical Trials—Patient-Reported Outcomes
The Reg-ent registry will soon be announcing opportunities for Reg-ent members and their practices to participate in research and clinical trial opportunities.
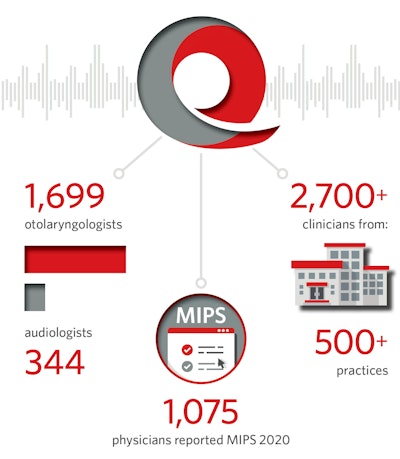
Reg-ent has been building its national otolaryngology-specific data repository for over six years. Available data covers the spectrum of disease states and procedures performed by otolaryngologist-head and neck surgeons and includes patient demographics, healthcare encounters, current and historical clinical problems, medications past and present, procedures performed, details on procedures performed, labs and imaging, lab orders, and patient notes. Reg-ent will soon include ancillary data like radiology orders and results, pathology, and audiogram data.
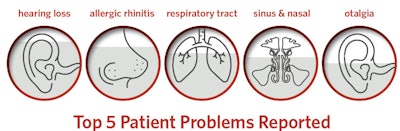
The clinical data in Reg-ent paints an interesting picture of larger trends from patient demographics to participating clinicians and primary diagnoses to locations. Hearing loss and allergic rhinitis are the top two diagnoses in Reg-ent.
Reg-ent’s patients are between the ages of 45 and 64, followed closely by the ages of 19 to 44 and with an almost equal female/male split.
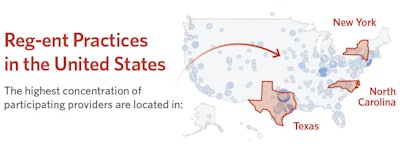
The three states with the highest number of participating clinicians are (in order) New York, Texas, and North Carolina. The states with the highest number of practices are (in order) Texas, California, and New York. Reg-ent includes both general otolaryngologists, as well as specialists in facial plastics, head and neck surgery, pediatrics, otology and neurotology, otolaryngic allergy and general allergy, and immunology. Care team members—including nurse practitioners, physician assistants, audiologists, and speech pathologists—round out the remainder of clinicians included in Reg-ent.
Driving Quality and Performance
Measuring Patient-Reported Outcomes
We are excited to present the first Reg-ent Patient-Reported Outcomes Module (PROM). Registry participants may utilize the survey instrument focused on shared decision-making in the treatment of age-related hearing loss in the Reg-ent dashboard, with the SNOT-22 survey next in line.
Reg-ent continues to support our members and their practices with 22 specialty-specific Qualified Clinical Data Registry (QCDR) measures (see pages 25-26) developed by, and for, AAO-HNS members for Merit-based Incentive Payment System (MIPS) reporting and quality improvement. Available in the Reg-ent dashboard, we ask everyone to assess these quality measures (labeled as AAO and as ASPS), review the measure specifications and workflows, and then select the measures that are most applicable to your providers and patients. QCDR measures are not limited to MIPS reporting but are to be utilized by all Reg-ent participants, regardless of practice setting and/or MIPS reporting requirements, for quality improvement purposes.
By including QCDR measures in your measures selection for 2021, and working on the mapping and validation processes, you will be providing otolaryngology-focused data to Reg-ent that will be used for measure refinement, new measure development, and will provide a foundation for research and practice improvement activities and initiatives. Submitting these measures for MIPS reporting also contributes to the creation of benchmarks for these measures and results in higher scores for these measures in future years of MIPS reporting.
EHR Solutions
A practice’s electronic health record (EHR) is a critical factor for successful participation in Reg-ent. Both the EHR vendor and hosting structure (on a local server in the practice or on a server in the 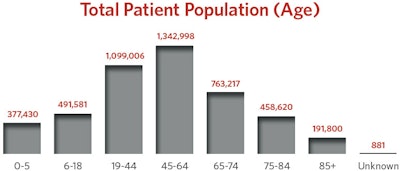
If your EHR solution is on a local server inside your practice, there generally are not fees associated with transferring your data. The data will typically be of higher fidelity as well, which helps build higher quality data in the registry. Cloud-based EHRs push data to Reg-ent, and several vendors with cloud-based solutions will charge the practice for such data transfer. Smaller, less well-known EHRs will sometimes lack resources and make practices bear the burden of data transfer.
EHRs that do not charge for data transfer and work well with Reg-ent include:
- Epic
- NextGen
- Medent
- Modernizing Medicine
The Reg-ent registry also works with:
- Waiting Room Solutions
- Greenway (locally hosted)
- Allmeds
- Aprima
- Nextech
- Medinformatix
- Medisoft Clinical
- Meditech
- Compulink
- eMDs
- Dr. Chrono
- PrognoCIS
- SRS Soft
The following cloud-based EHR vendors charge fees to practices that wish to push data to Reg-ent:
- Allmeds
- Allscripts
- eClinicalWorks
- CureMD
- Compulink
If you don’t see your EHR solution listed, please reach out to reg-ent@entnet.org.
Many practices assume that it is the Reg-ent registry that is problematic when, in fact, it is either the EHR solution or vendor that is problematic. Regardless, both Reg-ent and its vendor partner FIGmd continue to work with each EHR vendor to assure as seamless a process as is possible.











![05 Hpv Vaccination [converted]](https://img.ascendmedia.com/files/base/ascend/hh/image/2021/06/05_HPV_Vaccination__Converted_.60d0a1f25afe4.png?auto=format%2Ccompress&fit=crop&h=191&q=70&rect=45%2C0%2C1830%2C1830&w=340)