Providing Guidance and Expanding Research Capabilities
Research and Quality continues to support our volunteers in the development of guidance and expanding research capabilities as Reg-ent moves into Phase II.
 Cecelia E. Schmalbach, MD, MSc
Cecelia E. Schmalbach, MD, MSc
Coordinator, Research & Quality
Research and Quality continues to support our volunteers in the development of guidance and expanding research capabilities as Reg-entSM moves into Phase II.
All clinical practice guideline development, measures development, Reg-ent operations and data-curation initiatives, and CORE activities continued seamlessly thanks to our dedicated staff and membership who utilized virtual methods throughout 2020 and 2021. During this time period, we also supported the incorporation of guidance and research focused on COVID-19. The Patient Safety and Quality Improvement and Outcomes Research and Evidence-Based Medicine Committees each have contributed greatly to these efforts.

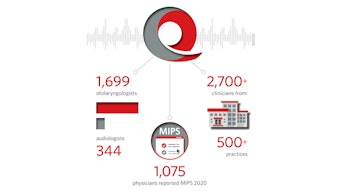
Measures staff responded in a proactive fashion to regulatory changes by the Centers for Medicare & Medicaid Services as outlined in the 2021 Medicare Physician Fee Schedule final rule. Public reporting will be transitioning from “traditional Merit-based Incentive Payment System (MIPS)” to MIPS Value Pathways (MVP). Our development efforts in this regard have positioned the membership well to be ahead of these changes, which are anticipated to begin as early as the 2022 reporting year.
More detail on research and quality initiatives is contained in the following articles highlighting important activities over the past year.










![05 Hpv Vaccination [converted]](https://img.ascendmedia.com/files/base/ascend/hh/image/2021/06/05_HPV_Vaccination__Converted_.60d0a1f25afe4.png?auto=format%2Ccompress&fit=crop&h=191&q=70&rect=45%2C0%2C1830%2C1830&w=340)