The Challenge of Screening for Head and Neck Cancer
Improving head and neck cancer screening should focus on refining risk-based approaches while integrating emerging technologies.
Madeline Dansky, BS, and Ameya A. Asarkar, MD, on behalf of the Head and Neck Surgery & Oncology Committee
Risk Factors
HNC arises from a combination of environmental exposures, viral infections, and host factors. Tobacco use remains the most significant risk factor for non–HPV-associated head and neck cancer and demonstrates a clear dose-dependent relationship.3 Alcohol consumption is an additional independent risk factor and acts synergistically with tobacco to significantly increase cancer risk.4
As tobacco use has declined in many developed countries, the epidemiology of HNC has shifted with HPV-associated disease emerging as the predominant cause of oropharyngeal cancer.4 High-risk HPV, particularly type 16, is strongly associated with oropharyngeal cancers and is more commonly seen in younger patients and nonsmokers.3 Additional risk factors include Epstein-Barr virus in nasopharyngeal carcinoma, poor oral hygiene, occupational exposures, nutritional deficiencies, and immunosuppression.
Key takeaway: Collectively, these factors contribute to chronic mucosal injury, predisposing patients to premalignant changes and increasing the risk of both primary and second primary tumors.
Red Flags
The early signs and symptoms of HNC are often subtle and nonspecific, contributing to delays in diagnosis. Clinical presentation varies widely depending on the anatomic site and etiology, and symptoms frequently overlap with those of benign conditions. Persistent symptoms lasting more than two to three weeks should raise concern for malignancy and warrant evaluation by a clinician. Common presenting features include a non-healing oral ulcer or mass. Dysphagia, odynophagia, and referred otalgia in the setting of a normal ear examination are also concerning and may reflect more advanced disease. Persistent voice changes, including hoarseness, are a classic early sign of laryngeal cancer, while progression may lead to dyspnea or airway compromise.
In HPV-associated disease, patients often present with painless cervical lymphadenopathy with minimal or absent primary site symptoms.5 Epstein-Barr virus–associated nasopharyngeal carcinoma may present with cervical lymphadenopathy, epistaxis, unilateral nasal obstruction, and, in more advanced cases, conductive hearing loss or cranial nerve deficits.5
Key takeaway: Given the variability and nonspecific nature of these presentations, a high index of suspicion is essential, highlighting the critical role of clinician awareness and screening in early detection.
Concept of Field Cancerization
Early detection is critical as survival outcomes are closely associated with stage at diagnosis. An important concept in the pathogenesis of head and neck cancers is “field cancerization.” Field cancerization refers to the widespread epithelial damage that occurs in the upper aerodigestive tract following chronic exposure to carcinogens such as tobacco and alcohol. Studies have shown that over time, this repeated injury leads to multiple independent areas of premalignant change rather than a single isolated tumor.5 As a result, patients are at risk not only for a primary tumor but also for synchronous and metachronous malignancies within the same region.
Key takeaway: This underscores the need for comprehensive evaluation and ongoing surveillance, as clinically normal-appearing mucosa may harbor early or premalignant disease.
Pitfalls in Head and Neck Cancer Screening
Screening for HNC presents several challenges, largely due to the lack of a standardized, effective screening modality. Unlike breast or colorectal cancer, there is no widely accepted screening test with sufficient sensitivity and specificity for early detection in asymptomatic individuals.
Although many cases present at advanced stages, the overall prevalence of HNC in the general population remains relatively low, limiting the utility of broad screening programs. Population-based screening may increase false positives, leading to unnecessary procedures, patient anxiety, and increased healthcare costs.1
Key takeaway: The anatomic complexity of the head and neck region can make early lesions difficult to detect, even with thorough examination. Differences in tumor biology between tobacco-related and HPV-associated cancers further complicate the identification of appropriate screening populations.5
Who to Screen
Screening efforts should focus on high-risk patients. This includes individuals with a history of HNC, premalignant lesions (such as dysplastic oral leukoplakia), and genetic predisposition syndromes (including Fanconi anemia, Li-Fraumeni syndrome, dyskeratosis congenita, and Plummer-Vinson syndrome).1 Patients with significant environmental or behavioral risk factors, such as heavy tobacco or alcohol use, should also be considered for closer surveillance.
Key takeaway: Recognition of these populations relies on targeted patient encounters, including detailed history-taking to assess risk factors. Routine examinations and repeat oral cavity inspections remain essential components of screening in these groups.
Tools for Screening
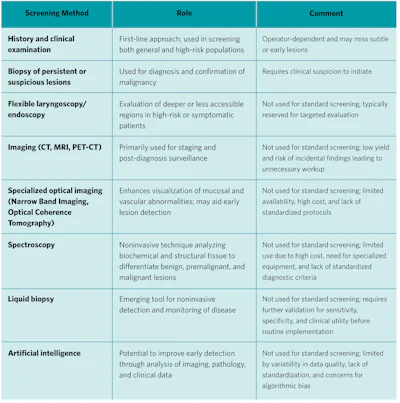
Screening begins with a thorough clinical examination, including visual inspection and palpation of the oral cavity, oropharynx, and larynx. Suspicious lesions should be biopsied for definitive evaluation. While this approach remains the standard, it is operator-dependent and may miss subtle or early lesions.
In high-risk patients or those with unclear presentations, flexible laryngoscopy or panendoscopy can be used to evaluate deeper regions of the upper aerodigestive tract and identify occult or second primary tumors. These modalities are typically reserved for high-risk populations due to cost and resource utilization considerations.
Imaging modalities such as PET-CT are primarily used for post-diagnosis surveillance to detect recurrence or metastasis rather than as primary screening tools. Other technologies, such as narrowband imaging (NBI), optical coherence tomography (OCT), and spectroscopy, have been explored for the early detection of head and neck cancers. NBI enhances visualization of mucosal and vascular patterns, allowing for identification of early neoplastic changes that may not be apparent on routine clinical examination.6 Spectroscopy-based technology has also been explored as an adjunctive tool for detection, utilizing biochemical and molecular changes associated with early malignancy.1 OCT is a noninvasive, high-resolution imaging technique used to visualize tissue microstructure and detect early neoplastic changes. These technologies are not used as primary screening methods due to limited cost-effectiveness and lack of standardization; they are best utilized as adjuncts to clinical evaluation in high-risk or symptomatic patients.
Overall, the evidence supports that screening is most effective when targeted to high-risk populations rather than applied broadly. A recent systematic review found that while screening may increase detection of early-stage HNC, evidence regarding its impact on survival, feasibility, and cost-effectiveness remains limited.7 Other research reinforces this approach, demonstrating that broad population-based screening for HNC has limited cost-effectiveness and supports the use of targeted, risk-based screening strategies.1
Key takeaway: Clinicians should maintain a low threshold for biopsy of persistent lesions and consider endoscopic evaluation in high-risk patients with nonspecific symptoms.
Emerging Technologies for Personalized Screening
Emerging technologies are expanding the HNC screening landscape. Liquid biopsy represents a promising, noninvasive method that detects tumor-related components in blood, saliva, or urine (including circulating tumor DNA and viral DNA from HPV and Epstein-Barr virus).3 In addition to early detection, it may also play a role in monitoring for recurrence. This approach remains investigational and is not yet the standard of care.
Artificial intelligence (AI) has also advanced screening capabilities by applying machine learning to imaging, genomic, and clinical datasets. These tools may improve the detection of subtle early malignancy and enhance risk stratification. Limitations include variability in data quality, lack of standardization, and potential algorithmic bias.8
Current Guidelines for HNC Screening
There is currently no recommended population-based screening strategy for HNC in asymptomatic adults. As a result, screening is focused on high-risk populations, including individuals with prior HNC, premalignant lesions, significant tobacco and alcohol use, HPV-related risk, and genetic predisposition syndromes. Screening primarily relies on clinical examination, including visual inspection and palpation of the oral cavity, oropharynx, and cervical lymph nodes.
Key takeaway: The U.S. Preventive Services Task Force has concluded that there is insufficient evidence to determine whether the benefits of routine oral cancer screening outweigh potential harms.9
Conclusion and Action Items
Future efforts to improve HNC screening should focus on refining risk-based approaches while integrating emerging technologies. Prioritizing high-risk populations may improve diagnostic yield and allow for earlier intervention.
Technologies such as liquid biopsy and artificial intelligence show promise for enhancing early detection but require further validation before routine implementation. Improving patient and clinician education remains critical to increase recognition of early lesions during routine medical and dental examinations.
From a public health perspective, increasing awareness of HPV-associated HNC and promoting vaccination remain essential prevention strategies. Continued research evaluating cost-effectiveness, diagnostic accuracy, and equitable implementation will be necessary to guide future screening recommendations and reduce head and neck cancer -related morbidity and mortality.
References
- Guntinas-Lichius O, Bücking C, Ng SP, et al. Targeted Screening Strategies for Head and Neck Cancer: A Global Review of Evidence, Technologies, and Cost-Effectiveness. Diagnostics (Basel). 2025; 15(16):2095. Published 2025 Aug 20. doi:10.3390/diagnostics15162095
- Ciortan Sirbu MI, Marin MA, Chioran D, et al. A Current Perspective of Two of the Most Aggressive Head and Neck Cancers: Pharyngeal and Laryngeal. Curr Oncol. 2025; 32(10):572. Published 2025 Oct 15. doi:10.3390/curroncol32100572
- Galati L, Chiocca S, Duca D, et al. HPV and head and neck cancers: Towards early diagnosis and prevention. Tumour Virus Res. 2022;14:200245. doi:10.1016/j.tvr.2022.200245
- Gormley M, Creaney G, Schache A, Ingarfield K, Conway DI. Reviewing the epidemiology of head and neck cancer: definitions, trends and risk factors. Br Dent J. 2022; 233(9):780-786. doi:10.1038/s41415-022-5166-x
- Johnson DE, Burtness B, Leemans CR, Lui VWY, Bauman JE, Grandis JR. Head and neck squamous cell carcinoma. Nat Rev Dis Primers. 2020; 6(1):92. Published 2020 Nov 26. doi:10.1038/s41572-020-00224-3
- van Schaik JE, Halmos GB, Witjes MJH, Plaat BEC. An overview of the current clinical status of optical imaging in head and neck cancer with a focus on Narrow Band imaging and fluorescence optical imaging. Oral Oncol. 2021;121:105504. doi:10.1016/j.oraloncology.2021.105504
- Sykes EA, Weisbrod N, Rival E, Haque A, Fu R, Eskander A. Methods, Detection Rates, and Survival Outcomes of Screening for Head and Neck Cancers: A Systematic Review. JAMA Otolaryngol Head Neck Surg. 2023; 149(11):1047-1056. doi:10.1001/jamaoto. 2023.3010
- Pham TD, Teh MT, Chatzopoulou D, Holmes S, Coulthard P. Artificial Intelligence in Head and Neck Cancer: Innovations, Applications, and Future Directions. Curr Oncol. 2024; 31(9):5255-5290. Published 2024 Sep 6. doi:10.3390/curroncol31090389
- U.S. Preventive Services Task Force. Oral cancer: screening. Published November 15, 2013. Accessed March 23, 2026. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/oral-cancer-screening