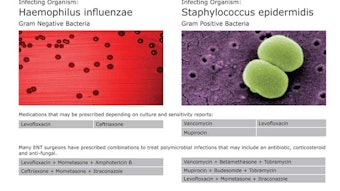
AAO-HNSF, AHNS Sponsor Thyroid Cancer Research
The incidence of thyroid cancer in the United States has more than doubled since the early 1970s. Between 1996 and 2005, the incidence rose annually by 5.8 percent among men and 7.1 percent among women, a more rapid increase than any other cancer site. This year the AAO-HNSF and the American Head and Neck Society (AHNS) are sponsoring two research projects aiming to increase our basic knowledge of thyroid cancer. The 2012 AHNS Alando J. Ballantyne Resident Research Pilot Grant was awarded to Neerav Goyal, MD, MPH, from the Pennsylvania State University College of Medicine in Hershey, PA, for his project entitled “Determining the Association between Radon Levels and Thyroid Cancer.” Exposure to radiation is one of the only known risk factors for developing thyroid cancer. However, in the vast majority of thyroid cancer patients, no such risk factor is ever identified. Radon is a radioactive colorless, odorless, and tasteless gas derived from the breakdown of uranium, which emits alpha particle radiation. Given the high rates of thyroid cancer in Pennsylvania relative to the rest of the United States, and the high levels of radon reported in many counties within Pennsylvania, Dr. Goyal and his team will look to determine if an association exists between radon levels and thyroid cancer, and also determine the strength of the association. The study will have three phases. First, the team will collect existing data on radon levels in Pennsylvania by geographic subunit. Secondly, the team will collect data on the incidence of thyroid cancer in these same subunits and determine if there is any correlation between the two variables using spatial analysis methods, such as the Moran I global measure and local indicator of spatial association tests. Thirdly, the team will compare areas by radon exposure and determine if there are significant differences in the characteristics of thyroid disease in the geographic subunits. Through this research, Dr. Goyal and his team hope to demonstrate a possible avenue of identifying a previously unidentified risk factor and preventing carcinomas of the thyroid. By shifting the paradigm from treatment to prevention, the research could provide a significant benefit to the general public. The proposed work is the first of its kind and innovative because it not only addresses the cause of an alarming trend of rising rates of thyroid cancer, but also may offer a solution and enable us to prevent further development of this disease. One of the 2012 AAO-HNSF resident research grants was awarded to Vlad C. Sandulache, MD, PhD, from Baylor College of Medicine in Houston, TX, for his project, “MRI-based Evaluation of Metabolic targeting in Anaplastic Thyroid Cancer.” Anaplastic thyroid cancer (ATC) accounts for about 50 percent of all thyroid cancer-related deaths. Current treatment paradigms rely on external beam radiation (XRT) as a primary treatment modality. As such, development of novel radiosensitizing regimens is crucial in the management of this deadly disease. XRT induces tumor cell death through the formation of reactive oxygen species (ROS), which cause DNA damage. Tumor cell resistance to XRT is driven in large part by the ability to generate sufficient reducing equivalents to neutralize ROS. Pharmacologic perturbation of metabolic pathways can decrease intracellular levels of reducing equivalents and potentiate ROS generation in response to XRT. Dr. Sandulache and his team have previously demonstrated that combining 2-deoxyglucose (a glycolytic inhibitor) with metformin (a mitochondrial respiration inhibitor) results in increased intracellular ROS levels and significant potentiation of XRT toxicity. To date, it has not been possible to evaluate the effects of this anti-metabolic regimen on tumor reducing potential in vivo. They propose to use real-time hyperpolarized (HP) magnetic resonance imaging (MRI) in the context of an orthotopic xenograft murine model of ATC to provide pharmacodynamic information meant to maximize radiosensitization. Using HP MRI they will optimize metabolic inhibition using the above mentioned agents and achieve maximal radiosensitization of ATC tumors. The study is expected to achieve two goals. First, it will begin to define a pharmacodynamic profile for metabolic inhibition, which can aid in the development of clinically relevant therapeutic regimens. Second, it will demonstrate that HP-MRI represents a suitable tool for evaluating the effects of metabolic targeting in this aggressive malignancy. Successful completion of this study will allow the team to draw meaningful conclusions regarding the validity of an anti-metabolic approach to radiosensitizing ATC tumors. Given the scarcity of available treatments for this deadly disease, any promising therapeutic regimen can significantly influence the current state of treatment. Although imaging is increasingly utilized in the discovery and staging of malignancy, it has rarely been employed to guide therapeutic intervention in real time. It is the team’s belief that an anti-metabolic strategy aimed at perturbing the tumor reducing potential can be facilitated by using HP-MRI in a manner that improves therapeutic effect. Using mathematical modeling, the group is currently developing new algorithms for the interpretation of HP-MRI data and adapting its utilization to multiple solid tumor models. The data obtained in this study are expected to contribute significantly to the development of these algorithms. In addition to providing novel pharmacodynamic information crucial to the development of new anti-metabolic agents, development of HP-MRI as a predictive tool will facilitate translation of treatment regimens used in this preclinical mode into clinical trials. Dr. Sandulache will conduct the above-described research under the mentorship of Stephen Y. Lai, MD, PhD, (Department of Head and Neck Surgery, University of Texas (UT) MD Anderson Cancer Center) and in collaboration with James A. Bankson, PhD, (Department of Imaging Physics, UT MD Anderson Cancer Center). Since 1997, more than $200,000 has been awarded to researchers through the Centralized Otolaryngology Research Efforts (CORE) grant program to improve our knowledge of thyroid cancer.
The incidence of thyroid cancer in the United States has more than doubled since the early 1970s. Between 1996 and 2005, the incidence rose annually by 5.8 percent among men and 7.1 percent among women, a more rapid increase than any other cancer site. This year the AAO-HNSF and the American Head and Neck Society (AHNS) are sponsoring two research projects aiming to increase our basic knowledge of thyroid cancer.
The 2012 AHNS Alando J. Ballantyne Resident Research Pilot Grant was awarded to Neerav Goyal, MD, MPH, from the Pennsylvania State University College of Medicine in Hershey, PA, for his project entitled “Determining the Association between Radon Levels and Thyroid Cancer.” Exposure to radiation is one of the only known risk factors for developing thyroid cancer. However, in the vast majority of thyroid cancer patients, no such risk factor is ever identified. Radon is a radioactive colorless, odorless, and tasteless gas derived from the breakdown of uranium, which emits alpha particle radiation. Given the high rates of thyroid cancer in Pennsylvania relative to the rest of the United States, and the high levels of radon reported in many counties within Pennsylvania, Dr. Goyal and his team will look to determine if an association exists between radon levels and thyroid cancer, and also determine the strength of the association.
The study will have three phases. First, the team will collect existing data on radon levels in Pennsylvania by geographic subunit. Secondly, the team will collect data on the incidence of thyroid cancer in these same subunits and determine if there is any correlation between the two variables using spatial analysis methods, such as the Moran I global measure and local indicator of spatial association tests. Thirdly, the team will compare areas by radon exposure and determine if there are significant differences in the characteristics of thyroid disease in the geographic subunits. Through this research, Dr. Goyal and his team hope to demonstrate a possible avenue of identifying a previously unidentified risk factor and preventing carcinomas of the thyroid. By shifting the paradigm from treatment to prevention, the research could provide a significant benefit to the general public. The proposed work is the first of its kind and innovative because it not only addresses the cause of an alarming trend of rising rates of thyroid cancer, but also may offer a solution and enable us to prevent further development of this disease.
One of the 2012 AAO-HNSF resident research grants was awarded to Vlad C. Sandulache, MD, PhD, from Baylor College of Medicine in Houston, TX, for his project, “MRI-based Evaluation of Metabolic targeting in Anaplastic Thyroid Cancer.” Anaplastic thyroid cancer (ATC) accounts for about 50 percent of all thyroid cancer-related deaths. Current treatment paradigms rely on external beam radiation (XRT) as a primary treatment modality. As such, development of novel radiosensitizing regimens is crucial in the management of this deadly disease. XRT induces tumor cell death through the formation of reactive oxygen species (ROS), which cause DNA damage. Tumor cell resistance to XRT is driven in large part by the ability to generate sufficient reducing equivalents to neutralize ROS. Pharmacologic perturbation of metabolic pathways can decrease intracellular levels of reducing equivalents and potentiate ROS generation in response to XRT.
Dr. Sandulache and his team have previously demonstrated that combining 2-deoxyglucose (a glycolytic inhibitor) with metformin (a mitochondrial respiration inhibitor) results in increased intracellular ROS levels and significant potentiation of XRT toxicity. To date, it has not been possible to evaluate the effects of this anti-metabolic regimen on tumor reducing potential in vivo. They propose to use real-time hyperpolarized (HP) magnetic resonance imaging (MRI) in the context of an orthotopic xenograft murine model of ATC to provide pharmacodynamic information meant to maximize radiosensitization. Using HP MRI they will optimize metabolic inhibition using the above mentioned agents and achieve maximal radiosensitization of ATC tumors. The study is expected to achieve two goals. First, it will begin to define a pharmacodynamic profile for metabolic inhibition, which can aid in the development of clinically relevant therapeutic regimens. Second, it will demonstrate that HP-MRI represents a suitable tool for evaluating the effects of metabolic targeting in this aggressive malignancy.
Successful completion of this study will allow the team to draw meaningful conclusions regarding the validity of an anti-metabolic approach to radiosensitizing ATC tumors. Given the scarcity of available treatments for this deadly disease, any promising therapeutic regimen can significantly influence the current state of treatment. Although imaging is increasingly utilized in the discovery and staging of malignancy, it has rarely been employed to guide therapeutic intervention in real time. It is the team’s belief that an anti-metabolic strategy aimed at perturbing the tumor reducing potential can be facilitated by using HP-MRI in a manner that improves therapeutic effect. Using mathematical modeling, the group is currently developing new algorithms for the interpretation of HP-MRI data and adapting its utilization to multiple solid tumor models. The data obtained in this study are expected to contribute significantly to the development of these algorithms. In addition to providing novel pharmacodynamic information crucial to the development of new anti-metabolic agents, development of HP-MRI as a predictive tool will facilitate translation of treatment regimens used in this preclinical mode into clinical trials. Dr. Sandulache will conduct the above-described research under the mentorship of Stephen Y. Lai, MD, PhD, (Department of Head and Neck Surgery, University of Texas (UT) MD Anderson Cancer Center) and in collaboration with James A. Bankson, PhD, (Department of Imaging Physics, UT MD Anderson Cancer Center).
Since 1997, more than $200,000 has been awarded to researchers through the Centralized Otolaryngology Research Efforts (CORE) grant program to improve our knowledge of thyroid cancer.