Charles W. Vaughan, MD—a Pioneer in Otolaryngology – ONLINE EXCLUSIVE
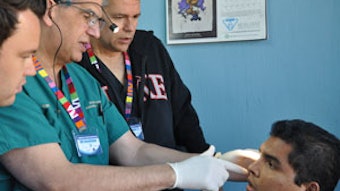
Louis Insalaco, MD Charles Williams Vaughan, MD, of Boston, who died in May, was a pioneer in otolaryngology. He was a clever innovator, a highly effective educator, and a superb mentor. A review of the contributions made by Dr. Vaughan parallels milestones in the development of modern otolaryngology. Dr. Vaughan worked with M. Stuart Strong, MD, physicists, and other clinicians to harness the carbon dioxide laser for use in otolaryngic surgery. Together they developed laryngoscopes and instruments to be used in microlaryngeal surgery. In a nearly uncanny kind of symmetry, Dr. Vaughan developed a tonsil malignancy late in his life that ordinarily would be treated with local surgical excision possibly combined with neck dissection and radiation therapy. However, always the innovator and unconventional thinker, Dr. Vaughan convinced his otolaryngologist, one of his former residents, to excise the tonsil cancer using a laser. Despite unconventional treatment, Dr. Vaughan remained free of tonsil cancer for the rest of his life. Around age 70, Dr. Vaughan became an amateur videographer and he captured for posterity chats with many of the foremost otolaryngologists of the modern era. When he was operating on a patient, Dr. Vaughan was performing several concurrent tasks; with his hands, he was completing the appropriate surgical procedure as he was demonstrating to those around him the meticulous technique required to achieve excellent results; simultaneously, he was considering ways to improve the procedure, and in his conversational patter he was prompting all around him to create innovations that could potentially benefit patients. There is much to be learned from the life and accomplishments of Dr. Vaughan.
Louis Insalaco, MD
 Charles W. Vaughan, MD
Charles W. Vaughan, MDCharles Williams Vaughan, MD, of Boston, who died in May, was a pioneer in otolaryngology. He was a clever innovator, a highly effective educator, and a superb mentor. A review of the contributions made by Dr. Vaughan parallels milestones in the development of modern otolaryngology. Dr. Vaughan worked with M. Stuart Strong, MD, physicists, and other clinicians to harness the carbon dioxide laser for use in otolaryngic surgery. Together they developed laryngoscopes and instruments to be used in microlaryngeal surgery.
In a nearly uncanny kind of symmetry, Dr. Vaughan developed a tonsil malignancy late in his life that ordinarily would be treated with local surgical excision possibly combined with neck dissection and radiation therapy. However, always the innovator and unconventional thinker, Dr. Vaughan convinced his otolaryngologist, one of his former residents, to excise the tonsil cancer using a laser. Despite unconventional treatment, Dr. Vaughan remained free of tonsil cancer for the rest of his life.
Around age 70, Dr. Vaughan became an amateur videographer and he captured for posterity chats with many of the foremost otolaryngologists of the modern era. When he was operating on a patient, Dr. Vaughan was performing several concurrent tasks; with his hands, he was completing the appropriate surgical procedure as he was demonstrating to those around him the meticulous technique required to achieve excellent results; simultaneously, he was considering ways to improve the procedure, and in his conversational patter he was prompting all around him to create innovations that could potentially benefit patients.
There is much to be learned from the life and accomplishments of Dr. Vaughan.