From the Education Committees: Facial Reinnervation
For patients with flaccid paralysis, the timing of intervention is critical because reinnervation options require the presence of viable muscle. Once denervated, muscle will begin to atrophy and fibrose after 12-18 months, and axons regrow at ~1 mm per day; therefore, even after a nerve repair is performed, it can still take months for the axons to reach the target motor endplates.
Marc Hale Hohman, MD, for the Facial Plastic & Reconstructive Surgery Education Committee
For patients with flaccid paralysis, the timing of intervention is critical because reinnervation options require the presence of viable muscle. Once denervated, muscle will begin to atrophy and fibrose after 12-18 months, and axons regrow at ~1 mm per day; therefore, even after a nerve repair is performed, it can still take months for the axons to reach the target motor endplates. For this reason, most surgeons will operate early whenever possible. Reinnervation is preferred over reanimation because the results appear more natural when the native facial musculature is used instead of placing an implant or transferring a muscle.
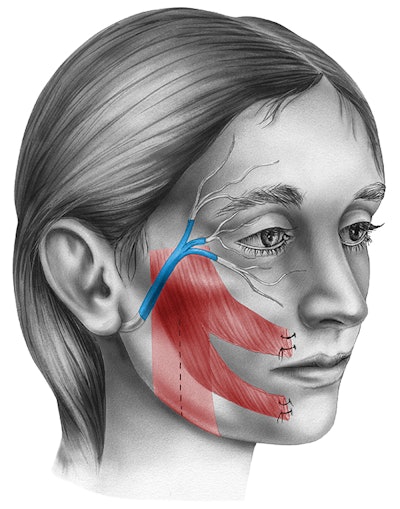
At its simplest, reinnervation is the repair of an injured nerve, whether a primary neurorrhaphy or cable graft, but it can also involve different types and combinations of nerve graft, depending on the specific deficits. Nerve injuries are ideally repaired within three days so that a stimulator can locate the distal stump before Wallerian degeneration is complete. More distal injuries have a better prognosis, and the small branches medial to the lateral canthus do not usually require repair to recover function. On the other hand, the more proximal the injury, the greater the likelihood of synkinesis. The fewer muscles the injured nerve branch innervates, the better the chance of a non-synkinetic recovery.
In cases with no viable proximal facial nerve, such as some acoustic neuroma patients, a nerve transfer may be required. There are many options, including the masseteric, hypoglossal, and contralateral facial nerve via cross face grafting. The masseteric nerve works well for smile rehabilitation because of its axon count, proximity to the recipient buccal facial nerve branch, and minimal donor deficit. The caveat to the masseteric nerve is that since the masseter muscle has low resting tone, the masseteric nerve does not provide much resting tone for the face, and is, therefore, better suited to restoration of voluntary movements, like smiling, particularly because masseteric nerve transfer patients need to bite down in order to smile. The hypoglossal nerve however does provide good resting tone and can also be used to restore voluntary movements. Unfortunately, loss of tongue function is more problematic than loss of the masseter; young patients may tolerate it well, but as they age, the oral deficit can become worse than the facial one would have been. Partial hypoglossal-facial transfers and jump grafts limit the donor deficit but also limit the potential for a good facial reinnervation.
Lastly, there are theoretical advantages of cross face nerve grafting, but results are inconsistent at best. The deficit from the donor nerves are minimal, both from the sural nerve graft harvest and from the donor buccal branch, and by connecting a buccal branch of the intact side to a corresponding branch on the paralyzed side, asynchronous and spontaneous smile should result because the patient does not have to bite or move their tongue to fire the nerve. Part of the problem may be a high rate of axonal loss across a long graft and two neurorrhaphies. Secondly, the cross face nerve graft is dependent on the function of the good facial nerve, which means that it is contraindicated in patients likely to develop a contralateral facial palsy, as in neurofibromatosis type 2. Since these donor nerves each have strengths and weaknesses, they are often used in combination to provide a synergistic result. A good example would be a hypoglossal-facial jump graft for resting tone with a masseteric nerve transfer for a smile and a cross face nerve graft for an eye blink. This would potentially restore resting tone and two volitional movements with minimal functional deficit or synkinesis.