Out of Committee: The future of vocal fold injectables
As the healthcare field continues to be shaped by the pressures of cost containment, efficiency, quality, and safety, otolaryngologists should expect to be influenced by the same factors. A key point where these forces intersect is in the management of vocal fold paralysis, particularly in the outpatient setting.
Edward Damrose, MD, Medical Devices and Drugs Committee
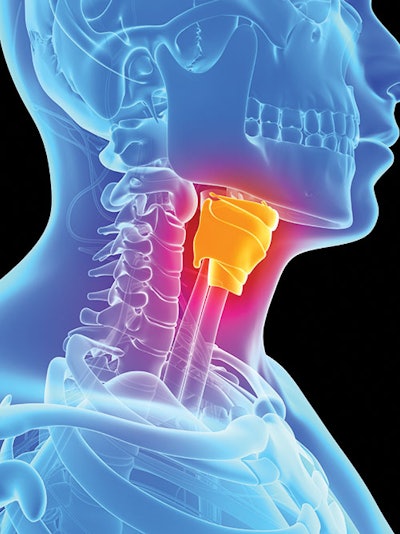
Vocal fold injection is a safe, cost-effective, and minimally invasive option in the treatment of vocal fold paralysis. A variety of materials have been described to treat this disorder, including fat, acellular human dermis, collagen, hyaluronic acid, calcium hydroxyapatite, and silicone, each with its own proponents and adherents.1
The ideal injectable would be biocompatible, easily injectable percutaneously or transorally, restore the normal viscoelastic properties of the vocal fold, nonreactive in host tissues, easy to adjust or remove in the event of inappropriate placement, “tunable” to the duration of effect needed, safe, and cheap. Unfortunately, the ideal injectable has yet to be created, and therefore, there continues to be ongoing research into new injectable materials. Advances in plastic surgery drive the majority of research and innovation in this field, as the most successful fillers, which are successful in the cosmetic arena, are often secondarily adapted for use in the laryngologic market. For the near future, it is likely that laryngology will continue to benefit from the economic incentives that drive innovation in the field of cosmetic surgery.
Paralysis and patient care settings
Surgery accounts for approximately 37 percent of patients with acute vocal fold paralysis and now surpasses extralaryngeal malignancy as the single leading cause of this disorder.2 Therefore, it is probable that otolaryngologists will become increasingly more likely to encounter this problem in acutely hospitalized patients over time. Vocal fold injection for acute paralysis, however, can safely and effectively restore glottic competence to this group of patients.3 Restoring glottic closure improves cough and pulmonary toilet, decreases aspiration, improves voice, and results in improved patient and referring provider satisfaction.3,4 The ability to transition a patient from tube feeding to oral feeding, shorten hospitalization, and decrease the need for adjunctive procedures and therapies, such as bronchoscopy, tracheostomy, or gastrostomy, can represent substantial cost savings for hospitals and patients’ insurers.
Vocal fold injection with materials such as calcium hydroxyapatite may afford similar voice outcomes to methods such as type one thyroplasty, in which a non-absorbable material is implanted adjacent to the paralyzed vocal fold to medialize it.5 Vocal fold injection is generally considered a temporary procedure, while thyroplasty is generally considered permanent. In its simplest and least expensive iteration, vocal fold injection can be done in the office or at the bedside and requires a flexible rhinolaryngoscope and monitor, the injectable, and topical anesthesia; injection under direct visualization using general anesthesia increases the cost. With thyroplasty, the procedure is more elaborate, requiring a complete sterile surgical set-up, intravenous sedation with monitored anesthesia care, and one or more hours of operative time.
Looking ahead
The last several years have seen the introduction of several new types of injectables as surgeons search for the ideal balance of durability, ease of injection, and concordance with normal vocal fold viscoelastic properties. Auricular cartilage and autologous platelet-poor plasma gel are host-derived and readily biocompatible.6-8 Hyaluronic acid, with or without cross-linked porcine collagen, is pre-mixed and has viscoelastic properties very similar to the normal superficial lamina propria.9,10 In step with biomaterial development, additional studies have continued to demonstrate the safety and efficacy of injection in the outpatient setting under simple topical or local anesthetic.11 Open surgical medialization has not seen as much development in this time period, perhaps as a consequence of these advancements. The introduction of preformed titanium implants, which afford simple and effective permanent vocal fold medialization, as well as novel devices that allow for easier arytenoid adduction, will likely join the open surgical options in the future.12,13
For patients who may require repeat injections over time, thyroplasty may prove to be a more economical long-term solution. Interestingly, however, up to two-thirds of patients who undergo early injection for acute vocal fold paralysis may avoid the need for later open correction, possibly because injection may assist the vocal fold in attaining a more functional final position in those patients who fail to recover complete mobility.14 These interesting findings generate new possibilities: perhaps injection may indeed be a cost-effective long-term solution to the problem of vocal fold paralysis. A timely push in the right direction may be what just what the doctor ordered for that troublesome hoarseness. While our field awaits the arrival of a new gold standard—perhaps the complete reversal of paralysis through neural regeneration or implantation of servomotors or pacers15—the need for improved vocal fold injectables will continue.
References:
- Damrose EJ, Berke GS. Advances in the management of glottic insufficiency. Curr Opin Otolaryngol Head Neck Surg. 2003 Dec;11(6):480-4.
- Rosenthal LH, Benninger MS, Deeb RH. Vocal fold immobility: a longitudinal analysis of etiology over 20 years. Laryngoscope. 2007 Oct;117(10):1864-70.
- Damrose EJ. Percutaneous injection laryngoplasty in the management of acute vocal fold paralysis. Laryngoscope. 2010 Aug;120(8):1582-90.
- Oestreicher-Kedem Y, Agrawal S, Jackler RK, Damrose EJ. Surgical rehabilitation of voice and swallowing after jugular foramen surgery. Ann Otol Rhinol Laryngol. 2010 Mar;119(3):192-8.
- Shen T, Damrose EJ, Morzaria S. A meta-analysis of voice outcome comparing calcium hydroxylapatite injection laryngoplasty to silicone thyroplasty. Otolaryngol Head Neck Surg. 2013 Feb;148(2):197-208.
- Lim YS, Lee YS, Lee JC, Lee BJ, Wang SG, Park HJ, Nam SB, Bae YC. Intracordal auricular cartilage injection for unilateral vocal fold paralysis. J Biomed Mater Res B Appl Biomater. 2014 Apr 25. doi: 10.1002/jbm.b.33189.
- Hong SJ, Lee SH, Jin SM, et al. Plasma gel as a new augmentation material for injection laryngoplasty in rabbit model: histologic analysis. Otolaryngol Head Neck Surg. 2013 Oct; 149(4):596-602.
- Woo SH, Kim JP, Park JJ, Chung PS, Lee SH, Jeong HS. Autologous platelet-poor plasma gel for injection laryngoplasty. Yonsei Med J. 2013 Nov; 54(6):1516-1523.
- Halderman AA, Bryson PC, Benninger MS, Chota R. Safety and Length of Benefit of Restylane for Office-Based Injection Medialization-A Retrospective Review of One Institution’s Experience. J Voice. 2014 Feb 13.
- Wen MH, Cheng PW, Liao LJ, Chou HW, Wang CT. Treatment outcomes of injection laryngoplasty using cross-linked porcine collagen and hyaluronic acid. Otolaryngol Head Neck Surg. 2013 Dec; 149(6):900-6.
- Powell J, Carding P, Birdi R, Wilson JA. Injection laryngoplasty in the outpatient clinic under local anaesthetic: a case series of sixty-eight patients. Clin Otolaryngol. 2014 Jun 13.
- Schneider-Stickler B, Gaechter J, Bigenzahn W. Long-term results after external vocal fold medialization thyroplasty with titanium vocal fold medialization implant (TVFMI). Eur Arch Otorhinolaryngol. 2013 May; 270(5):1689-1694.
- Hoffman HT, Heaford AC, Dailey SH, et al. Arytenoid repositioning device. Ann Otol Rhinol Laryngol. 2014 Mar; 123(3):195-205.
- Friedman AD, Burns JA, Heaton JT, Zeitels SM. Early versus late injection medialization for unilateral vocal cord paralysis. Laryngoscope. 2010 Oct;120(10):2042-6.
- Goldfarb D, Keane WM, Lowry LD. Laryngeal pacing as a treatment for vocal fold paralysis. J Voice. 1994 Jun;8(2): 179-85.