Early Hearing Aid Fitting to Protect Cognitive Health in Older Adults: A Geriatric Otolaryngology Perspective
Hearing loss and cognitive disorders in advanced years require a proactive approach centered on timely hearing aid fitting.
Francis Gutierrez Quintana, MD, and Carlos Takahiro Chone, MD, PhD, on behalf of the Geriatric Otolaryngology Committee
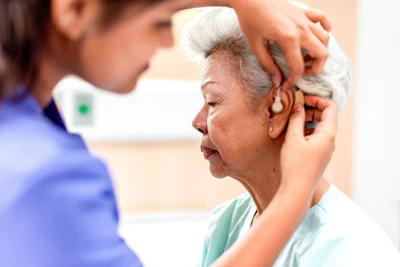
As populations mature worldwide, sensory aging has become a key determinant of functional independence, social participation, and emotional well-being. Hearing loss is a common sensory change in older adults and an important, potentially modifiable risk factor for cognitive disorders. Framing age-related hearing loss (ARHL) as a marker of both sensory and brain health highlights its significance beyond mere auditory impairment. This approach places otolaryngologists at the forefront of initiatives aimed at sustaining communication abilities, autonomy, and cognitive function among older adults.1,2,3,4
As the United States and numerous other countries face an unprecedented increase in their older populations, healthcare providers across all practice settings are encountering a greater number of elderly patients presenting with multiple coexisting health challenges—hearing loss, imbalance, visual difficulties, and cognitive concerns—all of which significantly affect quality of life and clinical outcomes. Epidemiological studies indicate that more than two-thirds of adults aged 70 and older experience at least mild hearing loss, and the likelihood of moderate-to-severe loss rises with advancing age. Major reports focused on dementia prevention have identified mid- to late-life hearing loss as a substantial contributor to preventable dementia cases, emphasizing auditory health as a high-yield target for preventive strategies.1,2,3
How Hearing Loss and Dementia Are Connected
ARHL reflects progressive damage along the auditory pathway, from cochlear hair cell and synaptic loss to central changes in temporal and spectral processing. These alterations degrade the clarity of the signal reaching the cortex, forcing older adults to recruit additional attention and working memory resources to follow everyday conversations, especially in noisy environments. Over the years, this chronic "effortful listening" may contribute to cognitive fatigue and unmask or accelerate underlying neurodegenerative processes.4,5,6
The link between hearing loss and dementia is likely multifactorial. Reduced auditory input may drive cortical reorganization and atrophy in auditory and associated networks. Hearing loss often leads to social withdrawal, loneliness, and depression—each independently associated with increased dementia risk. Shared vascular or neurodegenerative mechanisms may also underlie both ARHL and cognitive decline. Taken together, these mechanisms support the emerging view that treating hearing loss is not just symptom relief; it may be a meaningful way to modify cognitive trajectories in aging.2,4,5,6
What We See in Otolaryngogeriatrics Clinics
In geriatric otolaryngology practice, the clinical story is remarkably consistent: older adults report difficulty following conversations in restaurants or family gatherings, increasing reliance on others to repeat or "translate" what was said, and a gradual retreat from social, religious, or community activities. Family members frequently add concerns about forgetfulness, miscommunication, and tension around misunderstood medical instructions or financial decisions.1,4
In our university-based Otolaryngologic Geriatrics Clinic at IOU–UNICAMP in Campinas, São Paulo, Brazil—a public academic otolaryngology institute serving as a large and diverse reference center for the Brazilian public health system—we routinely see this overlap of hearing complaints, imbalance, visual strain, social withdrawal, and early cognitive concerns presenting at the same visit. Otolaryngologists, audiologists, geriatricians, and speech-language pathologists jointly evaluate communication, balance, mood, and cognition at each encounter. This reinforces a key message for Academy members: untreated hearing loss in older adults is rarely an isolated problem, and addressing it proactively can have meaningful ripple effects across multiple domains of healthy aging.3,4,7
Beyond Hearing: Balance and Vestibular Aging
Age-related sensory decline rarely affects just one system, and the vestibular apparatus is no exception. Vestibular hypofunction becomes increasingly common with aging and contributes to gait instability, falls, and reduced confidence in mobility, further limiting social engagement and physical activity. In older adults who already struggle to hear, the added burden of dizziness or unsteadiness can amplify isolation and reluctance to participate in group or community activities.3
At IOU–UNICAMP, a brief vestibular and balance assessment alongside the hearing evaluation helps identify patients whose communication difficulties are compounded by fear of falling and restricted mobility. For these individuals, coordinated management—including vestibular rehabilitation, fall-prevention counseling, and timely hearing aid fitting—can support both safety and the confidence needed to remain socially and cognitively engaged.3,7
Why Early Hearing Aid Fitting—within 90 Days—Matters
If hearing loss is a modifiable dementia risk factor, the timing of intervention may be just as crucial as the method used. Drawing on principles from stroke care, the concept of "time is brain" applies directly to ARHL: the longer an older adult experiences untreated, clinically significant hearing loss, the greater the cumulative impact of increased listening effort, social isolation, and sensory deprivation-induced changes within the brain.4,5
Emerging public health and geriatric literature supports early detection and management of ARHL as a strategy to reduce cognitive decline and dementia risk. Organizing practice workflows so that appropriate candidates move from diagnostic audiometry to hearing aid fitting within approximately 90 days is both biologically plausible and operationally achievable. A 90-day window helps prevent the slow, often invisible drift from a documented audiogram to "someday" amplification—minimizing delays that may otherwise lead to prolonged periods of effortful listening, social isolation, and avoidable cognitive deterioration.2,8
An early-fitting approach can:
- Shorten the period of intense listening effort by restoring audibility and improving speech understanding in everyday environments.9,10
- Help maintain social participation, family interaction, and engagement in cognitively demanding activities.1,8
- Provide an earlier opportunity to educate patients and caregivers about the relationship between hearing and cognition, promoting device adherence and realistic expectations.1,8
- Allow the care team to track hearing aid use, perceived benefit, and cognitive status over time, prompting timely referral for geriatric or neuropsychological evaluation when needed.9,10
At IOU–UNICAMP, we have begun treating the interval from diagnostic confirmation of significant hearing loss to effective amplification as a core quality indicator, aligning scheduling, audiology capacity, and counseling around that 90-day target. Similar time-based metrics can be readily adapted to diverse practice settings across the U.S.—from large academic centers to community clinics.3,7
Practical Steps for Academy Members
Even as high-level guidelines on hearing loss and dementia continue to evolve, several practical steps are already clear and actionable for otolaryngologists:
1. Explicitly frame age-related hearing loss as a modifiable risk factor for cognitive decline and discuss this with older patients and their families at every opportunity.1,2
2. Incorporate systematic screening for hearing loss and brief cognitive concerns into routine care for adults aged 65 and older, with special attention to those with moderate or greater audiometric loss.4,8
3. Work with audiology colleagues to establish a "90-day pathway" from diagnosis of clinically significant hearing loss to hearing aid fitting, and track this interval as a practice quality-of-care metric.8,9
4. Use follow-up visits to monitor hearing aid use, communication outcomes, and social participation, and to identify signs that warrant referral for geriatric or neuropsychological evaluation.9,10
5. Engage in interdisciplinary collaboration with primary care, geriatrics, neurology, psychology, speech-language pathology, and social work to support older adults facing both hearing and cognitive challenges.1,3,7
By treating auditory rehabilitation as a time-sensitive intervention for brain health—not merely a matter of comfort or convenience—otolaryngologists can help ensure that no older adult is "left unheard" at the very moment when communication, connection, and cognitive reserve matter most.2,4,10
Acknowledgments
The authors acknowledge the multidisciplinary team of the Otolaryngologic Geriatrics Unit at IOU–UNICAMP for their ongoing work in implementing and refining early-fitting pathways for older adults with hearing loss.
Disclosures
Conflicts of interest: The authors declare no financial or non-financial conflicts of interest related to the content of this article.
Funding: No external funding was received for the preparation of this manuscript.
AI tool usage: Artificial intelligence tools were used to assist in organizing and formatting portions of this manuscript. All content, clinical perspectives, and factual claims were authored and verified by the authors.
Author contributions: Both authors contributed substantially to the conception, drafting, and critical revision of this article and approved the final version for submission.
References
- Johns Hopkins Bloomberg School of Public Health. Hearing loss and the dementia connection. Published 2021. Accessed March 2026. https://publichealth.jhu.edu/2021/hearing-loss-and-the-dementia-connection
- Livingston G, Huntley J, Sommerlad A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. 2020;396(10248):413-446. doi:10.1016/S0140-6736(20)30367-6
- Marques LP, Conforto AB, Amaral B, et al. Self-reported hearing loss and associated factors in Brazilian older adults: a cross-sectional analysis of the EpiFloripa Aging Study. Cad Saude Publica. 2021. https://doi.org/10.1590/0102-311X00102420
- Albers MW, Gilmore GC, Kaye J, et al. Hearing loss and dementia in older adults: a narrative review. J Alzheimers Dis. 2024. PMID:38112446. doi:10.3233/JAD-231127
- Gurgel RK, Ward PD, Schwartz S, et al. Dementia and hearing loss: a narrative review. Maturitas. 2020;131:55-62. doi:10.1016/j.maturitas.2019.10.007
- Deal JA, Betz J, Yaffe K, et al. Hearing impairment and incident dementia and cognitive decline in older adults: the Health ABC Study. J Gerontol A Biol Sci Med Sci. 2017;72(5):703-709. PMID:27071780
- Buriti AKL, Gama-Chaves PR, Rodrigues-Oliveira MF, et al. The influence of cognition on the auditory skills of older people: pre- and post-hearing aid fitting. Audiol Commun Res. 2022. https://doi.org/10.1590/2317-6431-2021-2575
- Dawes P, Emsley R, Cruickshanks KJ, et al. Early detection and management of hearing loss to reduce dementia incidence. Age Ageing. 2025;54(1):afaf004. doi:10.1093/ageing/afaf004
- Maharani A, Dawes P, Nazroo J, Tampubolon G, Pendleton N; SENSE-Cog WP1 group. The effect of hearing aid use on cognition in older adults: a systematic review. Front Aging Neurosci. 2020;11:321. PMC7020090
- Mosner MG, Kinney AR, Kleindienst Robler S, et al. A comparative prospective longitudinal study of cognitive outcomes in older adults with hearing loss. Front Aging Neurosci. 2023;15:1302185. doi:10.3389/fnagi.2023.1302185






