Intranasal Epinephrine as a New Option for Anaphylaxis
With a needle-free alternative to intramuscular epinephrine administration that is less bulky to carry, we may see a paradigm shift in anaphylaxis management that could benefit our patients.
Haidy Marzouk, MD, MBA, on behalf of the Allergy, Asthma, and Immunology Committee
Food is the most common trigger for anaphylaxis episodes, and up to 8% of children in the U.S. have food allergies. There are approximately 125,000 food-induced allergic reaction emergency department visits per year in the U.S., 14,000 of which are due to anaphylaxis. One study found that adolescents aged 13 to 19 are three to four times more likely to die from anaphylaxis than children.2 Teenagers are at particular risk, with the peak incidence of deaths from anaphylaxis associated with peanut and tree nut allergy occurring in the 15- to 24-year-old age group.3 Many teenagers do not regularly carry their EpiPens or even know how to use them properly.1 Correct use of EA is surprisingly low, ranging from 16% to 32%, measured through both observational studies and patient reports. A recent meta-analysis of EA use found that the most common reasons for lack of use are availability, lack of education for parents/teens on how to administer epinephrine, concerns about systemic effects, misuse, and accidental administration.1 Certain factors have been shown to influence decision-making regarding EpiPens. The size or bulk of the device was noted in survey studies, which found that its size influenced respondents' decisions not to carry it all the time. Anxiety regarding needles and injections was present among several participants. Although fear of needles may impact usage, it was not necessarily shown to affect whether or not they carried the device.
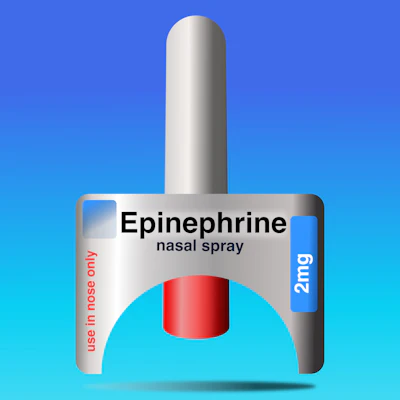
With these barriers present, a new option has come to the horizon for the treatment of anaphylaxis. On August 9, 2024, the FDA approved neffy® (epinephrine nasal spray). It is the first needle-free epinephrine formulation for the treatment of anaphylaxis in adults and pediatric patients weighing ≥30kg (2mg dose). In March of 2025, the indication was expanded to include patients ≥4 years old and weighing 15kg to less than 30kg, with a lower 1mg formulation. The approval was supported by four studies involving 175 adults that compared intranasal epinephrine with traditional epinephrine injection products. These studies showed comparable systemic epinephrine concentration and hemodynamic responses to traditional injectable treatment in multiple phase 1 and phase 3 studies. The time to maximum plasma concentration for neffy was similar to that of IM injection. Pediatric data showed similar plasma levels in children weighing ≥ 30kg. Other research, including repeat-dosing studies, has shown neffy’s pharmacokinetic profile to be consistent and tolerable across multiple administrations. Adverse events were generally mild. Preliminary observational data indicate an 89% success rate in treating anaphylactic episodes, comparable to injection response rates. The device is activated by inserting it into one nostril and pressing the plunger, without the need for priming. Despite its usability, administration errors remain a concern, as do those for IM epinephrine. These errors can be exacerbated in pediatric patients or those with cognitive or physical impairments.
The American Academy of Allergy, Asthma, and Immunology (AAAAI) emphasizes the need for clinician education and hands-on practice. Direct real-world data remained limited. Given that intranasal epinephrine is new to the market, the question remains whether IM epinephrine should still be available as an option, either primarily or as a backup for the treatment of anaphylaxis. Considerations for nasal administration include structural abnormalities that may affect mucosal absorption, local and systemic effects (e.g., nasal irritation, rhinorrhea, sneezing, throat discomfort, tachycardia, jitters), and coexisting conditions or medications. With a needle-free alternative that is less bulky to carry now available on the market, we may see a paradigm shift in anaphylaxis management that could benefit our patients. Awareness among our otolaryngology community, as well as our patient base, is crucial to improving outcomes related to epinephrine carriage and anaphylaxis treatment.4,5
References
- Glassberg B, Nowak-Wegrzyn A, Wang J. Factors contributing to underuse of epinephrine autoinjectors in pediatric patients with food allergy. Ann Allergy Asthma Immunol. 2021 Feb;126(2):175-179.e3. doi: 10.1016/j.anai.2020.09.012. Epub 2020 Sep 18. PMID: 32950683; PMCID: PMC7498408.
- Lockey R. Adolescents and anaphylaxis. Prim Care Respir J. 2012 Dec;21(4):365-6. doi: 10.4104/pcrj.2012.00090. PMID: 23090435; PMCID: PMC6548055.
- Macadam C, Barnett J, Roberts G, Stiefel G, King R, Erlewyn-Lajeunesse M, Holloway JA, Lucas JS. What factors affect the carriage of epinephrine auto-injectors by teenagers? Clin Transl Allergy. 2012 Feb 2;2(1):3. doi: 10.1186/2045-7022-2-3. PMID: 22409884; PMCID: PMC3299626.
- FDA Approves First Nasal Spray for Treatment of Anaphylaxis. FDA News Release. Fda.gov, August 2024.
- ARS Pharmaceuticals Announces FDA Approval of neffy® 1mg (epinephrine nasal spray) for Type I Allergic Reactions, Including Anaphylaxis, in Pediatric Patients Weighing 15kg to less than 30kg. Ir.ars-pharma.com, March 2025.