Nicaragua: Virtual Temporal Bone Dissection
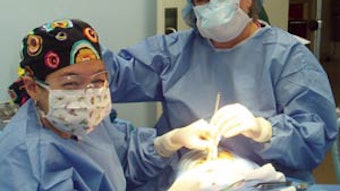
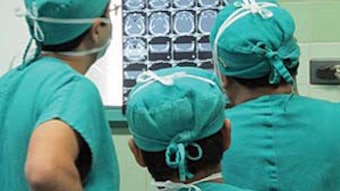
D. Richard Kang, MD, and Gregory J. Wiet, MDRichard Kang, MD, and Gregory J. Wiet, MD, two pediatric otolaryngologists from Nationwide Children’s Hospital and The Ohio State University, traveled to Managua, Nicaragua, to Escuela Hospital Antonio Lenin Fonseca in January. This was their fourth visit to Managua and this hospital that is the main institution for medical and surgical training in Nicaragua. They conducted an outpatient evaluation day and performed 13 surgeries including airway procedures, tympanoplasty, and mastoidectomy over the course of the next three days. Also, they were directly involved in training otolaryngology resident and attending physician training both through lectures on otology and pediatric airway topics and in the context of the surgical procedures performed. Unique to this visit was the performance of a “virtual” temporal bone dissection course. Drs. Kang and Wiet used a unique virtual temporal bone dissection system developed at Nationwide Children’s Hospital and The Ohio State University in conjunction with the Ohio Supercomputer Center under NIH funding (RO1 DC006458-01). The system makes use of a laptop computer with powerful graphics processing capability and a “haptic device,” which provides force feedback or a sense of touch, thus emulating the drill interaction with the temporal bone. This is an excellent example of how surgical simulation technology can greatly affect training in countries where traditional methods such as using cadaveric material are not available. Drs. Kang and Wiet plan to continue to return to Nicaragua on a regular basis to provide not only care for the population but training to the otolaryngology community for long-term impact.
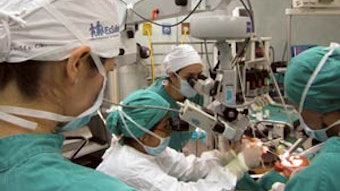
 Otolaryngology resident and attending physicians study temporal bone surgery using the Ohio State temporal bone simulator at Escuela Hospital Antonio Lenin Fonseca, Managua, Nicaragua.
Otolaryngology resident and attending physicians study temporal bone surgery using the Ohio State temporal bone simulator at Escuela Hospital Antonio Lenin Fonseca, Managua, Nicaragua.D. Richard Kang, MD,
and Gregory J. Wiet, MDRichard Kang, MD, and Gregory J. Wiet, MD, two pediatric otolaryngologists from Nationwide Children’s Hospital and The Ohio State University, traveled to Managua, Nicaragua, to Escuela Hospital Antonio Lenin Fonseca in January. This was their fourth visit to Managua and this hospital that is the main institution for medical and surgical training in Nicaragua.
They conducted an outpatient evaluation day and performed 13 surgeries including airway procedures, tympanoplasty, and mastoidectomy over the course of the next three days. Also, they were directly involved in training otolaryngology resident and attending physician training both through lectures on otology and pediatric airway topics and in the context of the surgical procedures performed. Unique to this visit was the performance of a “virtual” temporal bone dissection course. Drs. Kang and Wiet used a unique virtual temporal bone dissection system developed at Nationwide Children’s Hospital and The Ohio State University in conjunction with the Ohio Supercomputer Center under NIH funding (RO1 DC006458-01). The system makes use of a laptop computer with powerful graphics processing capability and a “haptic device,” which provides force feedback or a sense of touch, thus emulating the drill interaction with the temporal bone. This is an excellent example of how surgical simulation technology can greatly affect training in countries where traditional methods such as using cadaveric material are not available.
Drs. Kang and Wiet plan to continue to return to Nicaragua on a regular basis to provide not only care for the population but training to the otolaryngology community for long-term impact.