Crowdsourcing knowledge: Learning from the Patient Safety Event Reporting Tool
We all have a wealth of experiences that guide our individual medical wisdom and decision-making process. How much more could our collective wisdom help us all troubleshoot and improve processes in our individual practice environments? There is much to be learned from our combined experiences.
Michael E. McCormick, MD, and C.W. David Chang, MD, for the Patient Safety and Quality Improvement (PSQI) Committee
We all have a wealth of experiences that guide our individual medical wisdom and decision-making process. How much more could our collective wisdom help us all troubleshoot and improve processes in our individual practice environments? There is much to be learned from our combined experiences.
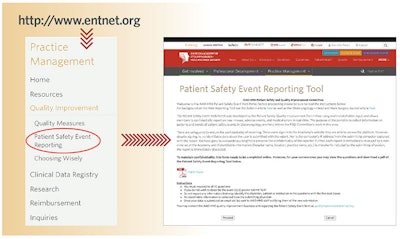
In 2014, the American Academy of Otolaryngology—Head and Neck Surgery (AAO-HNS) launched a web-based Patient Safety Event Reporting Tool (PSERT), accessible through the Academy’s website www.entnet.org with member login. This reporting tool was modeled partly after a similar system used by the Federal Aviation Administration and was intended for use both as a resource for Academy members to learn from others’ experiences and as a tool to identify developing trends. Digital safeguards have been designed in the database to ensure anonymity.
The AAO-HNS Patient Safety and Quality Improvement (PSQI) Committee helped develop and test the platform prior to launch. The first report of the project published by Vila et al in 2017, reviewed 53 events collected from its inception January 7, 2014, to October 7, 2015. The authors reported that most entries resulted in harm to the patient, and the most common events were technical errors.
A more recent review of the database performed by the PSQI Committee evaluated cases through December 2016. There was a cumulative total of 88 separate reports entered into the portal. Like the previous progress report, most cases resulted in harm to the patient, and most occurred in the operating room or in the hospital. One-third of entered cases involved an additional procedure for the patient and 11 percent involved the death of the patient. Interestingly, almost 60 percent involved what could be classified as a known sequelae of a procedure. Fourteen entries reported system or process errors, and 14 included defensive language or placed blame on another individual or a piece of equipment.
Subspecialty breakdown of reported cases reveals that most cases involved head and neck surgery (25), followed by neuro- otology (11), and laryngology (11). This is, of course, not an indicator for frequency of events in our subspecialties.
“Big data” collection initiatives such as Reg-entSM, the American College of Surgeons National Surgical Quality Improvement Program, and Medicare/insurance claims data are systematic and broad in their collection of data. However, these databases are unable to capture situational detail, system/process issues, and communication factors that inform our understanding of safety events. In contrast, PSERT intentionally queries physicians for these details, without which root cause analysis is difficult.
Qualitative rather than quantitative analysis is the objective. The intent is neither to statistically measure the incidence of nor derive the statistical significance of otolaryngic complications. Rather, the tool allows members to share knowledge and learn from each others’ events. While the reporting of any patient safety issue is welcome, we especially encourage sharing events that highlight:
- Medical/surgical technique problems
- System/process issues
- Knowledge gaps
- Issues requiring further attention by AAO-HNS leadership
The fruits of this project require voluntary contributions by physicians from all practice types: academic and private, solo and group, rural and urban, and civilian and military. Practices big and small review patient safety issues in a variety of ways. Hospital-based and academic-based practices have traditionally held “Morbidity and Mortality” conferences. Many smaller practices hold safety and quality reviews routinely during business meetings.
To facilitate data collection and reporting of events, physicians can download and print out a hard copy of the PSERT questionnaire located on the PSERT landing page. Having the printed form is helpful to structure discussion during safety and quality conferences and/or to ensure that proper richness of detail is collected regarding each event discussed. Use of the form can be customized to the needs of each organization. For example, physicians could utilize the form to submit events for discussion. Alternatively, the form could be used as a structured way to take minutes. Either way, data collected on the form can later be transferred to the online database.
The PSQI Committee will periodically review PSERT data and report to the AAO-HNS membership through both summative articles and case-specific articles written in the Bulletin to highlight areas of interest and need. Information will also be shared with the Education Committees to provide feedback for physician-reported knowledge gaps so that educational offerings can be created to address deficiencies.
One recent interesting case from PSERT featured a pediatric tracheostomy tube that was inserted upside down in surgery. The Bivona® brand tube is flexible throughout its cannula shaft. Prior to insertion of the trach, a rigid obturator with a blunt-tipped end is inserted in the flexible trach shaft to provide some rigidity during placement. In this instance, the obturator was inserted into the trach upside down (rotated 180 degrees). As the pediatric Bivona trach shaft is flexible, the tube conformed to the curve of the misinserted obturator. The trach was inserted into the patient (upside down). The mal-intubation was discovered post-operatively after ventilation difficulties were encountered. The tube was removed, rotated 180 degrees, and reinserted with good ventilation ability. This case provided feedback and education to the operating room staff, was applicable to all facets of the procedure, and identified areas for study and improvement.
The PSERT has proven an effective tool for the Academy membership to report adverse events. Your continued participation and contributions will help strengthen its utility as we all work together toward the common goal of improving patient safety. We need YOU to contribute and share with us your experiences. For more information, please contact us at qualityimprovement@entnet.org.
References
1. Vila PM, Lewis S, Cunningham G, et al. A Novel Patient Safety Event Reporting Tool in Otolaryngology. Otolaryngol Neck Surg. 2017;157(1):117-122. doi:10.1177/0194599817700363.